 
To help show this three-dimensional shape even more accurately, we can rely on space-filling models as well as ball-and-stick models. Seesaw molecular geometry: Disphenoidal or Seesaw (also known as Sawhorse is a type of molecular geometry where there are four bonds to a central atom with. We will discuss the significance of these electrons at the end of this section. The two dots above nitrogen indicate a lone pair of electrons that are not involved in any covalent bond. 1 This is one geometry for which the bond angles surrounding the central atom are not identical (see also pentagonal bipyramid ), because there is. Sample Problem 8-17 Predicting Dipole Moments Which molecule has a dipole moment, CIF, or XeF4. In chemistry, a trigonal bipyramid formation is a molecular geometry with one atom at the center and 5 more atoms at the corners of a triangular bipyramid. However, in the more detailed structural formula on the right, we have a dashed line to indicate that the rightmost hydrogen atom is sitting behind the plane of the screen, while the bold wedge indicates that the center hydrogen is sitting out in front of the plane of the screen. The Molecular Science John Olmsted, Gregory M. In the structural formula to the left, we are only seeing a two-dimensional approximation of this molecule. Keep in mind, however, that atoms and molecules, just like everything else in the universe, exist in three dimensions-they have length and width, as well as depth. 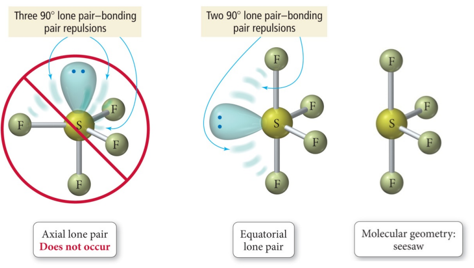 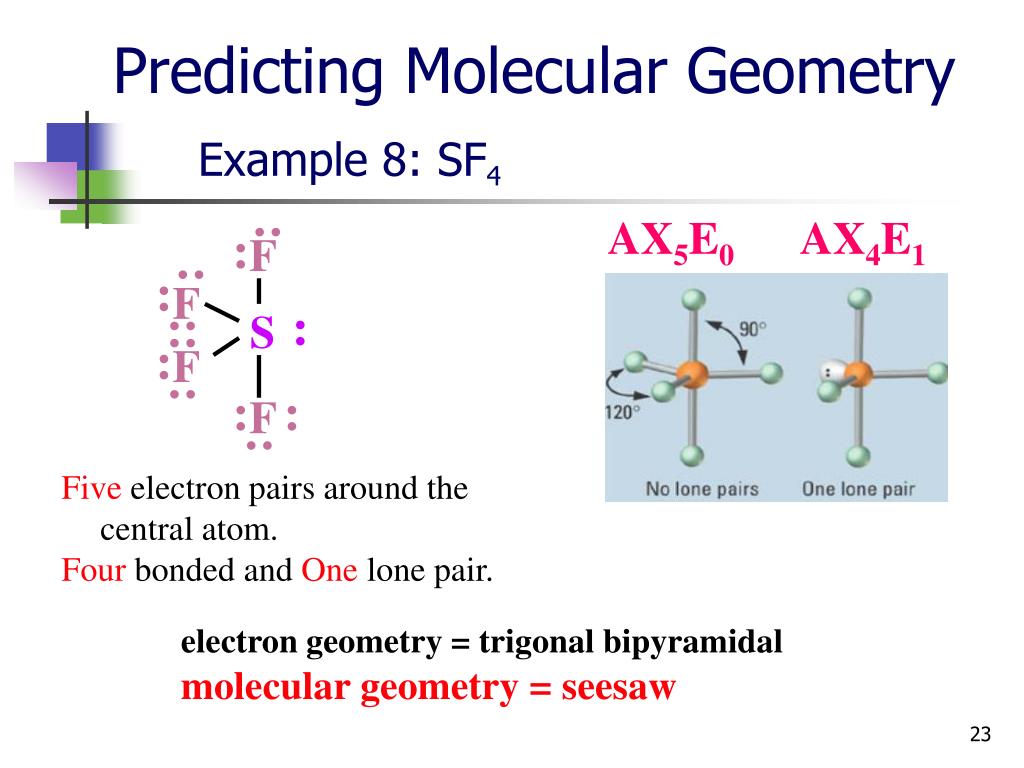
From both of these structural formulas, we can see that the central nitrogen atom is connected to each hydrogen atom by a single covalent bond. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
